The Nobel Prize in Medicine or Physiology in 2015 was awarded for two pharmacological discoveries: Tu Youyou’s discovery of the efficacy in malaria of qinghao, Artemisia annua, the source of artemisinin-based treatments, and the discovery of the avermectin group of macrolide antimicrobial drugs by Satoshi Ōmura and William C Campbell.

Figure 1. Two of the three Nobel Prize winners for medicine or physiology in 2015 (photo credits: Bengt Nyman – Own work, CC BY-SA 4.0; Ōmura; Campbell)
Ōmura had isolated the actinomycete Streptomyces avermitilis (previously NRRL 8165) at the Kitasato Institute, from a soil sample collected at Kawana, Ito City, in the Shizuoka Prefecture, Japan. He and his colleagues at the Kitasato Institute, and others at Merck Sharp & Dohme’s Research Laboratories in Rahway, New Jersey, to whom they sent their samples, extracted compounds from it and called them avermectins. One of them, abamectin was marketed as a treatment for equine parasites, and another, ivermectin, was marketed as Mectizan for human infections.
It was Campbell at the Merck Institute for Therapeutic Research who recognized the efficacy of ivermectin against the onchocerciasis parasite Oncocerca cervicalis, and this is now one of its main uses. It is also used for strongyloidiasis and scabies.
Two weeks ago, in discussing articles that were retracted after publication, I quoted some comments on a discussion website that had greeted a preprint that described apparently strikingly beneficial effects of ivermectin in covid-19. They illustrated the types of often uncritical responses that work of that sort attracts. This one particularly caught my attention:
○ It’s somewhat common, we still don’t know a definitive mechanism of action for Tylenol. There are many other medications with unknown MoA. Am a pharmacist. |
It is disappointing that a pharmacist should appear insouciant about the fact that an apparently dramatic piece of clinical evidence should not be supported by mechanistic evidence. Now it is true that in the Oxford Levels of Evidence mechanistic reasoning is regarded as the lowest form of life in the evidence menagerie. However, as I have pointed out elsewhere, with detailed reasons, this is a category error. Mechanistic reasoning is not itself evidence, although it should be backed up by strong evidence if it is going to be convincing. It is also true that there are treatments that we use regularly whose mechanism of action is unknown. Nevertheless, when faced with a myriad of choices for a new acute disease, a supporting mechanism can be a guide.
The mechanism of action of ivermectin in parasitic infections is thought to be inhibition of anion flux through a glutamate-controlled chloride channel present in the parasites, killing them by neurotoxicity. But the channel is not found in humans. Nor in viruses. Mechanisms, however, can always be conjured up. Here’s one from the same thread of comments:
|
This, perhaps convincing sounding mechanism, based on some in vitro evidence in other viruses, has been dusted off, with apparent authority, to explain the action of ivermectin in covid-19. But we have no evidence that this proposed mechanism is at all relevant (Figure 2). In fact, it can’t be relevant, because (spoiler alert) SARS-CoV-2 doesn’t enter the nucleus to replicate; it replicates in the cytoplasm.
If ivermectin is at all effective, an action on an importin of any kind isn’t the mechanism. Most of the time when we choose a drug to test, we seem to be entering a dark covid-19 tunnel. Is ivermectin the light at the end of the tunnel, or is it, as so often seems to be the case, just the light of a train coming in the other direction?
As ever, we are reduced to mouthing the same old trite truism, that we must wait for the results of well designed trials, preferably double-masked and placebo-controlled.
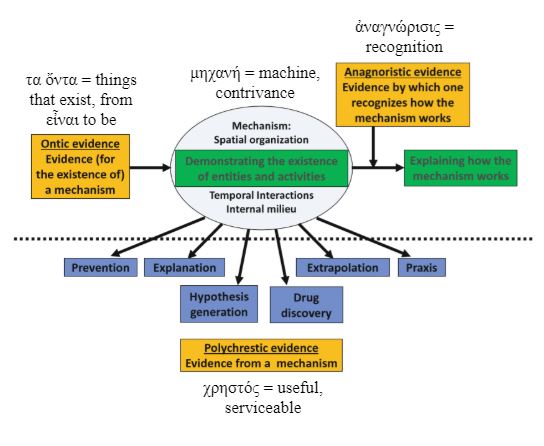
Figure 2. Different types of mechanistic evidence and the Greek origins of the names I have given them; for a satisfactory explanation of the action of any investigative compound we need three types of evidence before we should be thinking about a clinical trial in covid-19:
(a) evidence that the mechanism exists in SAR-CoV-2 (evidence for the mechanism or ontic evidence)
(b) evidence about how the mechanism would work and how the drug would affect it (anagnoristic evidence), and, optionally,
(c) evidence that the mechanism is likely to be relevant to the clinical problem (evidence from the mechanism, polychrestic evidence)
Absence of (a) and (b) should make us think twice about doing a clinical trial
Jeffrey Aronson is a clinical pharmacologist, working in the Centre for Evidence Based Medicine in Oxford’s Nuffield Department of Primary Care Health Sciences. He is also president emeritus of the British Pharmacological Society.
Competing interests: None declared.

