The Philippines moved quickly to expand COVID-19 testing capacity, but much of this new capacity remains idle. Emphasis must now shift to increasing productivity, particularly by improving the flow of samples to the laboratories.
Preceding entry: Scaling Up Capacity for COVID-19 Testing in the Philippines
When the first COVID-19 case was detected in the Philippines in February 2020, only the national reference laboratory had testing capability. The Department of Health (DOH) developed a four-pronged scale up strategy with technical support from ThinkWell Philippines to confront this challenge: enhancing capacities of sub-national laboratories, licensing more testing centers, setting up big testing centers and pop-up laboratories, and integrating innovative testing approaches — supported by the private sector, through the formation of Taskforce T3 (Test, Trace, Treat). With 57 licensed public and private facilities as of 15 June 2020, a rated testing capacity of 50,060 tests per day was estimated across 15 of 17 regions in the country.
Why is the system underperforming?
Data shows capacity is not used effectively. The average daily testing output is only 21% of the rated capacity, with 79% left unused (Figure 1). Several issues contributing to this have been identified.
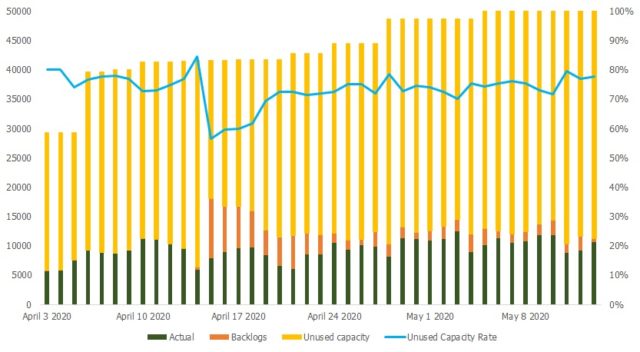
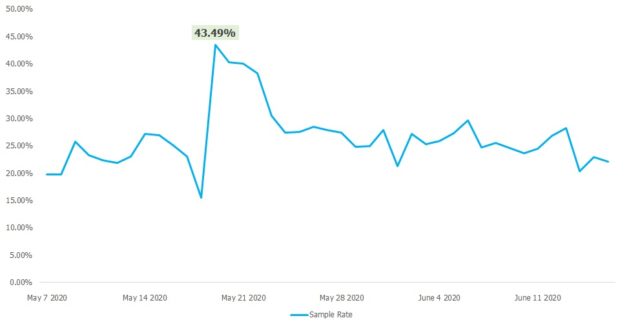
- Fewer specimens collected than rated capacity. The average samples laboratories receive for testing daily is only 26.42% of their rated capacity. On some days, some facilities do not report any samples at all. The highest number received for testing was last 19 May 2020, but was only 43.49% (23,519) of the rated capacity then.
- Some facilities receive too many samples, resulting in backlogs. On the average, 84% of daily samples are tested, 16% form part of backlogs. These are observed mostly in facilities in urban areas where population and cases are typically higher.
- Facilities have also reported a halt in operations on some days due to depletion of consumables. The lack of visibility on supply levels and needs at the laboratories also make procurement and distribution unresponsive.
- The testing algorithm is not regularly updated to reflect increased testing capacity. The DOH initially set very specific priorities for testing (Figure 3). This was to balance rational use of limited supplies with ensuring that those who needed to be tested are prioritized.
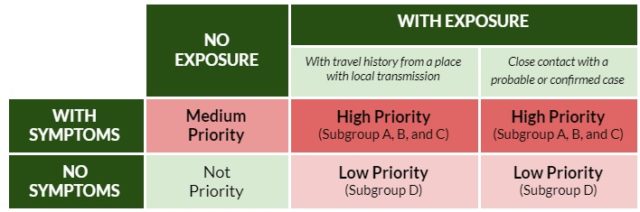
As more facilities were licensed, strict adherence to this meant the number of samples collected was less than the overall testing capacity.
Turning the Theoretical into Actual
The high capacity for testing only translates to benefits if actual tests are conducted. Several interventions are being undertaken to achieve this.
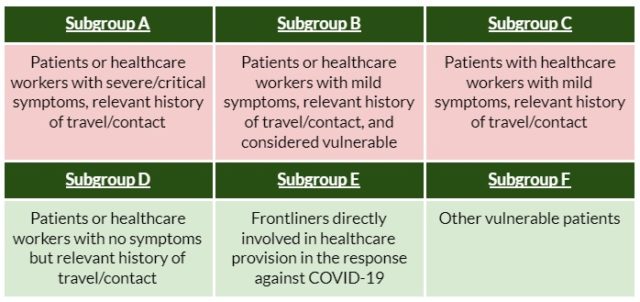
- Increasing specimen collection to maximize testing capacity. The DOH has expanded its algorithm for testing to include more population groups (Figure 4) – specifically frontline workers and vulnerable patients. The new guidelines indicate that around 20-30% of daily capacity of sub-national laboratories should be allocated for testing of health workers. Big swabbing centers have been put up to facilitate more sample collection. Local governments are engaged to implement and link these with contact tracing.
- Ensuring supplies for uninterrupted operations. The DOH, with support from Taskforce T3, developed an automated supplies inventory management system that conveniently collects real-time inventory data from all testing centers. Supplies, particularly test kits, are centrally procured to leverage scale. These ensure prompt mobilization of resources to avoid backlogs and operational disruptions. This is also the first system that allows DOH to track commodities down to facility level real time.
- Expanding zoning guidelines for better distribution of samples. Initial guidelines of the DOH only cover the first nine testing centers, assigning them catchment areas for testing. The new zoning guidelines set region-formed referral networks, linking testing centers and specimen collection sites within and across nearby provinces. This aims to improve traffic of samples, ensure faster turnaround times, and lessen backlogs.
Lessons Learned
Stemming the spread of the pandemic begins with extensive testing. Collaborative public-private work has led to increased capacity for testing in the Philippines, building greater confidence among the population. The new DOH target of 1.5M tests by the end of July 2020 means an average of 22,000 to 25,000 tests per day must be conducted from beginning of the month. While this is well within capacity, interventions across other components in the testing process should be improved, particularly specimen collection and stability of supplies.
To do this, government response needs to be more proactive than reactive. The automated inventory system is a step towards the right direction, and can enable efficient response even as demand increases. A pragmatic approach, coupled with timely and accurate data, is also crucial to ensure adoptable and applicable interventions. The stronger engagement with local governments in expanding swabbing sites and establishing zoning areas are good steps forward, and should continue to be a primary focus.
About the Authors
Christian Edward Nuevo1, Jemar Anne Sigua1, Kim Patrick Tejano2, Samuel Quizon3, Jessa De Guzman3, Maria Eufemia Yap1
- Strategic Purchasing for Primary Health Care (SP4PHC), ThinkWell Philippines
- Health Regulations Team, Department of Health Philippines
- Public Health Services Team, Department of Health Philippines
Declaration: Authors declare no competing interests.