Intraoperative neurophysiological monitoring (IONM) is generally used during complex surgeries that can compromise central and peripheral nervous system structures, providing real-time information about the functional integrity of these pathways and detecting reversible neurophysiological changes. This is particularly relevant in the surgery of intramedullary spinal cord (SC) tumors, which is associated with a high risk of morbidity. Therefore, the use of monitoring and mapping techniques of the corticospinal tract (CT) in such cases are crucial. However, mapping techniques of the CT at the SC are less developed, with additional concerns that motor responses may be evoked after stimulation of the dorsal column (DC), potentially producing false localization of the CT in the context of distorted anatomy.
In the July issue of JNNP, Deletis and colleagues describe a new neurophysiological approach to differentiate the CT from DC during the resection of intramedullary SC tumors. A prospective cohort study was performed in 32 patients undergoing SC tumor surgery. Mapping of the SC (i.e. from the surface or inside the resection cavity) was performed with electrical stimulation using a bipolar concentric probe. Motor evoked potentials (MEPs) were recorded from upper and lower limb muscles bilaterally. Overall, MEPs were obtained in 94% of the patients after the stimulation of the CT and 80% after the stimulation of the DC. Interestingly, MEPs triggered after DC stimulation had a longer latency and smaller amplitude than those triggered after CT stimulation, suggesting different lengths of the respective pathways. Moreover, after using a double train stimulation paradigm (with an intertrain interval of 60 ms), the stimulation of the CT elicited two identical MEP responses, whereas DC stimulation elicited only one response in patients without spasticity and a second MEP response that was always different from the first in patients with spasticity.
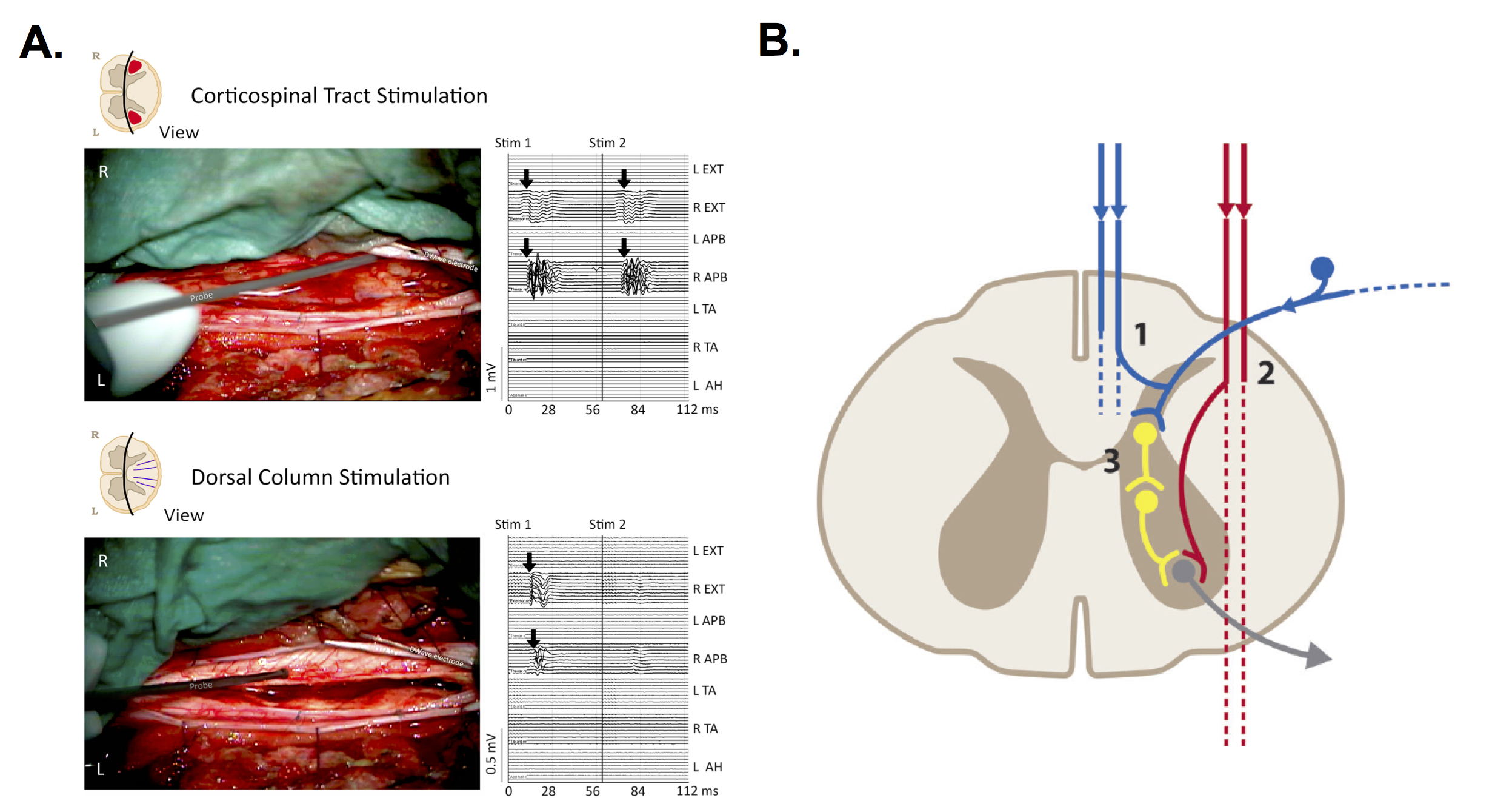
The current study presents a novel neurophysiological mapping technique of the CT and DC by direct electrical stimulation. By using a double train stimulation paradigm the authors demonstrate MEPs features that can clearly distinguish between CT and DC tract stimulation. Physiologically, the longer recovery time after DC stimulation and the complexity of the pathways support this finding. It has also been suggested that a “centrally activated H reflex” may explain the generation of MEP responses after DC stimulation (see figure 1). From a clinical and surgical point of view, the differentiation of the CT from the DC has important practical implications, given that a false positive MEP could confuse the surgeon about the localization of the CT, resulting in either motor deficits or incomplete tumor resection. Further studies are needed to confirm these findings, as well as assess the utility of motor mapping techniques in SC tumours. In the meantime, optimization of IONM protocols and exploration of novel electrophysiological techniques should be perused.
Read more at https://jnnp.bmj.com/content/89/7/754