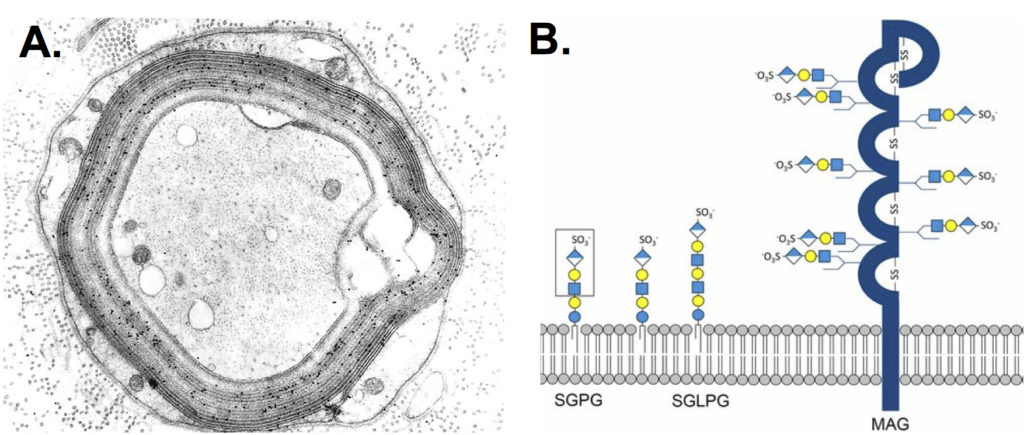
Neuropathy associated with anti-myelin-associated glycoprotein (anti-MAG) IgM antibodies typically presents as a distal variant of chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) called distal acquired demyelinating symmetric (DADS) neuropathy. Clinically, it is characterized by chronic distal symmetric sensory or sensorimotor deficits, associated with sensory ataxia and postural tremor. However, patients with positive anti-MAG antibodies range from oligosymptomatic patients to patients with proximal and distal impairment which emulates the “classical” CIDP phenotype. Moreover, for a therapeutic point of view, DADS seems to be refractory to immunomodulatory strategies, independently of the presence of anti-MAG antibodies (Fig.1).
In the April issue of JNNP, Svahn and colleague report findings from a multicenter study characterizing the clinicopathological phenotype and the response to treatment in patients with neuropathy associated with anti-MAG antibodies. The relationship of these variables with antibody titer was also assessed. Using a retrospective and prospective approach the authors analyzed standardized reports and medical records of 202 patients from 14 neuromuscular centers. The antibody titers were classified as having low (≥1000 to <10000 Bühlmann Titre Units; BTU), medium (10000–70000) or high (≥70000) anti-MAG antibodies. In summary, the mean age at disease onset was 62.6 years (25–91.4) and the antibody titers were low, medium or high in 11%, 51% and 38% of patients respectively. Sixty–eight per cent of patients presented with a monoclonal gammopathy of undetermined significance. Interestingly, from a clinical point to view 17% of patients had an atypical clinical presentation, which included acute or chronic polyradiculoneuropathies or multifocal neuropathies, independently of anti-MAG titers. Regarding therapeutic strategies, 78% of patients received immunotherapies. Most of them received rituximab in monotherapy or in combination with other immunosuppressants. Clinical response with rituximab was observe in 31.5% of patients and correlated with antibody titers ≥10000 BTU. Finally, responder patients (at 7–12-month follow-up), presented shorter disease duration in comparison with non-responders, though this comparison was non-significant after logistic regression (3.6 years (SD 3,7] vs 5.1 years (SD 4,2], p=0.06).
This current study presents the largest cohort of patients with anti-MAG antibodies published in the literature to date. Of relevance, the study evidences the variable clinical phenotype in patients with anti-MAG neuropathy, which interestingly is independent of antibody titers. That is, patients with low anti-MAG titer may have a ‘typical’ DADS phenotype. Even though two controlled studies failed to show the benefit of rituximab as monotherapy in primary endpoints, the present study suggests a beneficial use of rituximab in early stages of the diseases. There is no doubt that this information will have clinical implications in the development of future clinical trials in anti-MAG neuropathy.
Read more at http://jnnp.bmj.com/content/89/5/499