In many cases in medical care the systems are inefficient; time is wasted because of a myriad of processes and protocols and a failure to recognise the causes of delay in these processes. Dr. Douglas Woodhouse is a physician-engineer and a healthcare consultant focusing on process improvement. Here he describes a new way to improve wait times.

Dr. Douglas Woodhouse is a physician-engineer and a healthcare consultant focusing on process improvement
Cycle time is the total time that it takes from beginning to end of a treatment process, including treatment of complications. Efficiency rises when cycle times are short because care has been provided in less time, using fewer resources. Because the impact of adverse events is included, it is also an indicator of quality. Cycle time should be adopted as a key indicator of healthcare performance, and in this article I describe how this may be done.
Long wait times for health services are a concern in many health systems. However, it is likely that some efforts to reduce wait times have actually increased the time that it takes to complete treatment. Accepting more new patients into a healthcare process (reducing the wait time) without increasing the capacity of the process merely shifts the patient backlog from before the first to the second appointment.
Because most diseases lead to progressive disability, delays in completing treatment are not just inconvenient, but lead to worse health outcomes and higher costs. In comparison to measuring wait times, or length of stay and readmission rates, cycle time is a superior measure of overall healthcare performance.
Nine Rules for Low Cycle Time
1. Cycle time must include complications.
If a hospital just measured cycle time from admission to discharge there would be incentives to skip steps, discharge people home too early, and just deal with problems later on.
The solution is to include the management of complications in the cycle time. Cycle time for surgery would start at the time of referral and end when the patient is discharged home post-surgery. If the patient developed a post-operative complication, the cycle time clock would be re-started.
If cycle time includes treatment of complications, then it is safe and appropriate to reduce cycle time as much as possible. The shortest possible cycle time can only be achieved by providing optimal care.
2. The constraint should be patient intake.
The constraint (or “bottleneck”) is the slowest process step. Wait lists develop before the constraint.
If patient intake is the constraint, then the number of patients starting and completing treatment are matched. Wait times do not grow inside the process and cycle time is minimised.
If the wait list at intake increases, then the capacity of the entire process must be increased (rather than just accepting more patients at intake and hoping for the best).
3. Cycle time must be regularly monitored.
If cycle time increases then wait times inside the process should be measured to find the constraint (not only the wait time before intake, but also wait times before other activities). Wait times should be measured as patient-days. Either a small number of patients with long waits, or a large number with short waits indicates a constraint that should trigger action.
4. Once the constraint has been identified, that activity should be sped up.
Once the constrained process step is sped up, the constraint will shift to a different step. Each constraint within the process should be identified and eliminated (until the constraint moves to intake, where it belongs).
5. Process steps should be allowed to idle.
In a process that is constrained at intake, all other steps have extra capacity. Process steps will have idle time while waiting for a patient to complete the prior step. This is acceptable, and even desirable, as long as cycle time is not increasing.
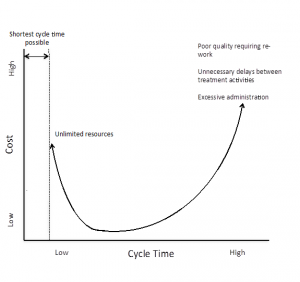
The shortest possible cycle time is defined by the minimum time required to provide all medically necessary services. Although in general shorter cycle times save money, the minimum cycle time can only be achieved if there are no resource limitations, and therefore relatively high costs. The lower left segment of the curve therefore represents the highest value (short cycle time, low cost and high quality).
6. Process steps should be completed in parallel whenever possible.
Parallel process steps prevent unnecessary waiting for sequential treatment activities and therefore significantly reduce cycle time.
7. Treatment should be as standardised as possible.
At least 80% of patients should progress through the standard process steps. This means that no more than 20% of patients should receive non-standard, individualised treatment. Time that is gained (through greater efficiency) by treating the majority of patients in a standard process can be used for the minority of patients who require extra care.
8. Protocols should be followed for the standard treatment process.
Protocols reduce the variability of the process (and therefore reduce the cycle time). The standard protocol should apply to the 80% of patients that follow the standard process. On the other hand, patients that are not in the standard process should not necessarily be treated according to a protocol.
9. The most expensive process step may justify the creation of a buffer (wait list) to ensure that it is always in use.
The most expensive process step should be maximally utilised. To achieve this, a short buffer (wait list) may be created to compensate for variations in capacity and demand. The maximum wait list for this step should be clearly defined.
If this maximum is exceeded, no additional patients should be accepted into the process until the cause of the delay is identified and fixed.
An Example of Cycle Time in the Operating Theatre
Dr Doug Woodhouse will be participating in a BMJ Quality Webinar on the 15th January from 5:00 – 6:00 (GMT+0). The session will take place through the WebEx platform at quality.bmj.com.